AGEM-102
Cell-Based Immunotherapy Reimagined
Overview
AGEM-102 is a next-generation engineered mesenchymal stem cell therapy developed using TrafEn. It is designed to deliver combinatorial therapeutic payloads directly to the tumour microenvironment, enabling localized and coordinated therapeutic activity.
By integrating precise cellular engineering with a unique dual-prong immunomodulatory and chemotherapeutic approach, AGEM-102 represents a scalable and versatile cell-based approach to targeted cancer therapy.
Why it matters
- Tumour-homing cell therapy:
- Dual-action mechanism:
- Non-viral innovation:
- Engineered for precision:
- Off-the-shelf ready:
- Engineered MSCs act a biological delivery vehicles, homing to cancer sites for localized, sustained action.
- Harnesses the synergy of targeted chemo- and immune- activation without systemic overload.
- Avoids mutagenic risks and vector size limits; enables high-yield payload delivery.
- Viral vectors are messy and random. AGEM-102 uses non-integrative, targeted transfection, avoiding genomic disruption while preserving cell function.
- Cryopreservation-compatible for scalable deployment.
How it works
AGEM-102 is an engineered mesenchymal stem cell (MSC) therapy developed using TrafEn, AGEM Bio's proprietary non-viral gene delivery platform. TrafEn enables MSCs to co-express cytosine deaminase (CDUPRT) and interferon-beta (IFNb), programming the cells to function and localized therapeutic biofactories within the tumour microenvironment.
AGEM-102 leverages the natural tumour-tropic properties of MSCs to preferentially home to tumour sites. Once localized, CDUPRT coverts 5-fluorocytosine (5-FC) into the toxic chemotherapy drug 5-fluorouracil (5-FU) directly within the tumour vicinity, while IFNb provides complementary anti-tumour and immunomodulatory activity.
By confining therapeutic activity to the tumour microenvironment, AGEM-102 is designed to enable targeted cancer killing while limiting systemic exposure and reducing treatment-related toxicity.

Oncology developmental pipeline

Real-life benefits
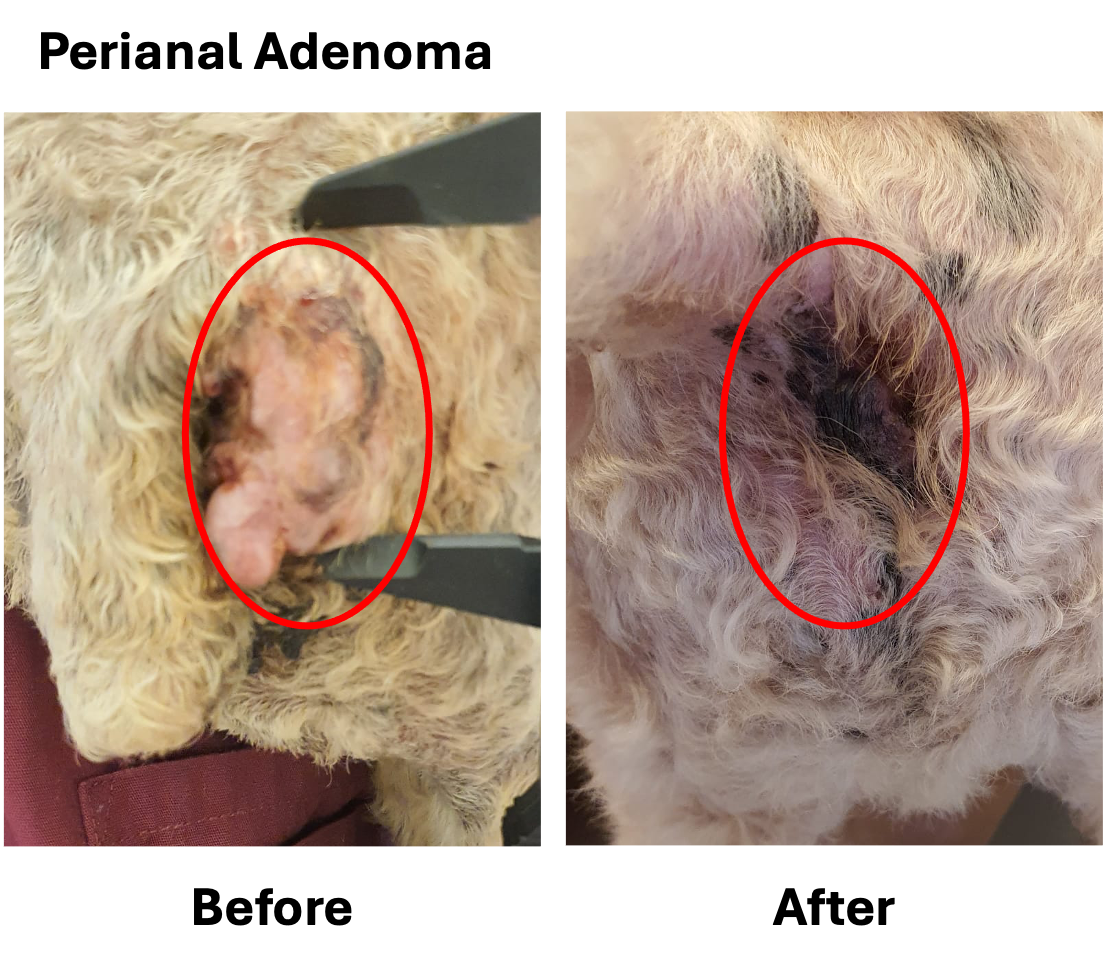
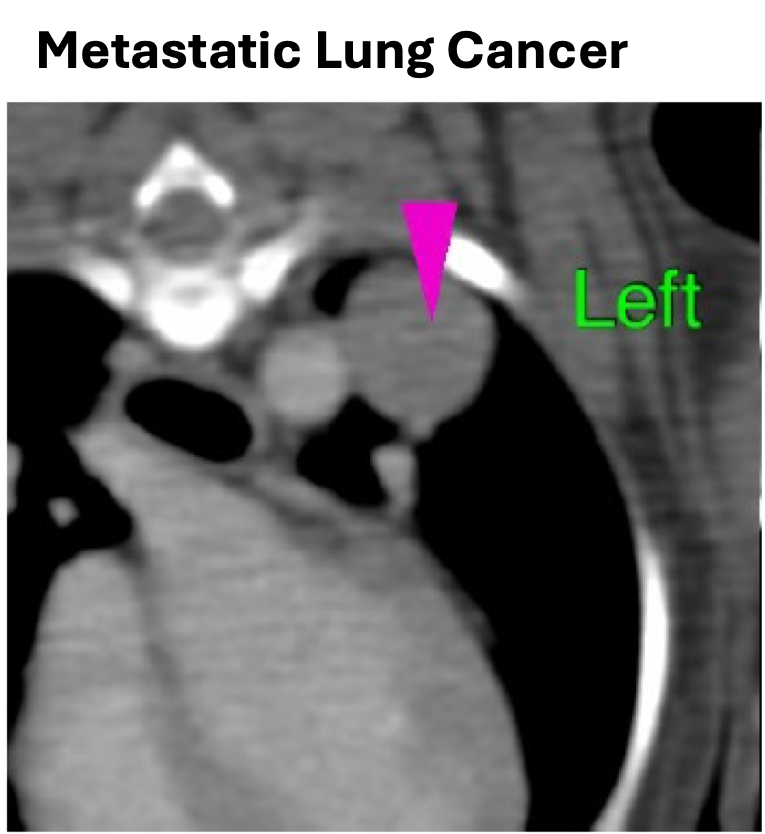
Veterinary medicine serves as a key translational pathway for advancing AGEM Bio’s engineered mesenchymal stem cell (MSC) platform toward human clinical applications. Through VM-102, our veterinary translational program, we apply the same non-viral genetic engineering principles that underpin our human pipeline to address serious, treatment-refractory diseases in companion animals.
VM-102 is designed for dogs and cats with advanced cancers where standard options are limited or exhausted, providing access to innovative, cell-based therapies while generating clinically relevant insights. By leveraging TrafEn-enabled MSC engineering, VM-102 enables controlled delivery of defined therapeutic factors in naturally occurring disease settings that closely mirror human pathology.
Importantly, data and experience gained from veterinary applications help de-risk and refine our engineered MSC platform, strengthening its development for human oncology and other indications. This integrated translational approach accelerates innovation while maintaining a clear focus on advancing transformative therapies for human patients.